 
Consult specific manufacturers for their guidelines. Suggested molar ratios of fragments to inserts are dependent on the size of the fragments and how many fragments are being assembled. 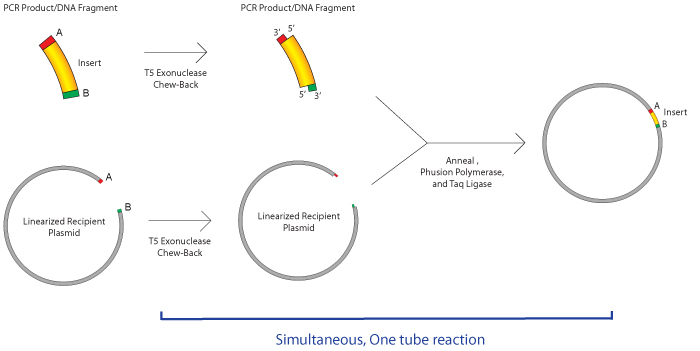
Determine the concentration with UV spectroscopy. If additional bands are present, you should purify your insert from an agarose gel. If only your expected PCR product is present, you can purify the PCR product away from primers, dNTP’s and residual enzyme with a PCR clean-up column. QuantitationĪfter amplification, check your PCR products on an agarose gel. The overlapping regions selection box from SnapGene. When designing your cloning project, you can imagine that your primers have two distinct components, the target-specific primer for amplification and the 5’ tail that will create the overlap between the vector or adjacent fragments. The length of the homologous ends depends on the number of fragments being assembled and their length (see below). PCR primers need to be designed to amplify your fragment of interest and include 20 or more base pairs of homology to the vector or adjacent fragments in your cloned product. ► Learn to simulate Gibson Assembly in SnapGene Insert Preparation You have the option to prepare your vector by linearizing your plasmid with restriction enzymes or by using Reverse PCR. Insert fragments are always prepared by PCR. To perform Gibson assembly, you will need to prepare one or more inserts and your vector. This article refers to all these reactions generically as Gibson Assembly. Second-generation assembly reactions such as HiFi Assembly and Gibson Assembly Ultra improve on these limitations. The classic Gibson Assembly reaction can result in errors at fragment junctions and does not work with single-stranded fragments or short fragments. The reaction is designed to occur at 50☌, with all enzymes selected for their stability and activity at this temperature. T5 exonuclease chews back the 5’ end of double-stranded DNA to expose engineered overlaps. Polymerase begins filling in at the overhangs preventing excessive enzyme chewing. When stable compatible overhangs anneal, ligase completes the fusion of the DNA fragments. Once ligated, the DNA is protected from the exonuclease. DNA primers are far more stable and easier to store, and they require less hard-to-come-by enzymes to initiate synthesis (see Chapter 2, Figure 1).These enzymes work together to fuse overlapping DNA fragments. Scientists use DNA primers instead of RNA primers for a variety or reasons. Living organisms solely use RNA primers, while primers used in the lab are usually DNA primers. What types of primers are there? RNA vs DNA primers The main property of primers is they must be complementary to the DNA template strand, serving to “prime” the strand for DNA polymerase to bind to and initiate DNA synthesis. Primers can also be called oligonucleotides and are literally small pieces of single-stranded nucleotides, generally about 5 – 22 base pairs in length. Primers are simple but key ingredients for DNA synthesis both within our bodies and within scientific experiments. Once primers are designed, run in silico PCR, or use them to plan critical tasks such as restriction cloning, Golden Gate assembly, and Gibson cloning. Be able to easily attribute results from experiments with the exact set of primers used, or see which sequences a primer is associated with. Link primer information directly in the Benchling Notebook and Benchling Registry providing full traceability for every experiment where a primer was used. With Benchling, teams can easily access shared primer libraries, upload new primer sequences, or design brand new primers. Primers are key ingredients in DNA synthesis, a process that occurs in sequencing, cloning, PCR, and other molecular biology methods in the lab. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
